|
 |
| J Korean Soc Geriatr Neurosurg > Volume 18(1); 2022 > Article |
|
Abstract
Idiopathic hypertrophic spinal pachymeningitis (IHSP) is a rare condition characterized by chronic progressive diffuse inflammatory fibrosis in the dura mater of the spinal cord. Depending on its extent or severity, IHSP may cause symptoms by compressing the spinal cord. We present a case of IHSP across the cervical dura mater in a 78-year-old male patient. The patient visited the emergency department for right shoulder pain a month before admission, weakness in the right upper extremity a week before admission, and sudden quadriparesis on the day of admission. A neurological examination of the motor power of the extremities indicated grade IV quadriparesis and grade I hand grasp power in both hands. Hypoesthesia under the T1 dermatome was evaluated. Cervical magnetic resonance imaging (MRI) with gadolinium enhancement showed diffuse thickening of the anterior and posterior dura through the C2 to C5 levels, causing central canal stenosis and compressive myelopathy. Immediate decompressive laminoplasty was performed. A biopsy specimen of the thickened dura mater was obtained during surgery, and IHSP was diagnosed on the basis of the results of the pathological examination. High-dose steroid therapy was administered after decompressive surgery, and follow-up MRI showed radiological improvement in the lesion area.
Idiopathic hypertrophic spinal pachymeningitis (IHSP) is a very rare disease first described by Charcot in 1869 [1]. IHSP presents as a progressive diffuse inflammatory fibrosis that occurs in the dura mater of the spinal cord and mainly affects the cervical and thoracic regions [2]. Progressive thickening of the dura mater causes compression of the spinal cord or nerve, and depending on the extent or degree of compression, myelopathy, radicular pain, and radiculopathy may appear [3].
Contrast-enhanced spinal magnetic resonance imaging (MRI) is a common imaging examination to diagnose IHSP, and the final diagnosis is made through excision biopsy of the dura mater of the relevant area [4]. On MRI, the characteristic findings of IHSP include hypo-intensity on an intradural extramedullary mass on T1- and T2-weighted images. In the case of involvement of more than 2 vertebral segments, the contrast enhancement pattern may appear different depending on the degree of peripheral active inflammation and central fibrosis [5,6]. The characteristic pathological findings of IHSP include chronic nonspecific granulomatous inflammation with fibrosis of the dura [7].
Although studies on treatment and long-term prognosis remain lacking, immunosuppressive therapy using steroids is the main treatment and is known to help improve symptoms [8]. Surgical decompression is required in patients showing no response to medical treatment or those showing neurological symptoms due to spinal cord compression [9].
A 78-year-old male patient was admitted to our hospital with quadriparesis that occurred on the day of admission. The patient had been experiencing posterior neck pain and right shoulder pain since approximately 2 months, and paralysis of the right upper extremity appeared one week before admission, for which he was admitted to another hospital. The patient was taking medications for hypertension and diabetes mellitus and had no specific infectious history, including tuberculosis. His family history showed no notable features.
Neurological examination showed that the patient was alert with no significant deficits in the cranial nerve examination. The motor power of the extremities indicated grade IV quadriparesis, and hand grasp strength was grade I for both hands. Hypoesthesia under the T1 dermatome level was checked. Deep tendon reflexes of the extremities were normal, and the anal and bladder tone were intact. On the basis of these findings, we diagnosed symptoms of cervical myelopathy and conducted additional tests.
A blood test was performed, and both C-reactive protein and erythrocyte sedimentation rate values were increased, suggesting inflammation. The tumor marker test did not show any specific findings, and acquired immunodeficiency syndrome, syphilis, tuberculosis, and other infections could be excluded through lab findings. A cerebrospinal fluid (CSF) test was performed, and leukocytosis with predominantly lymphocytes was confirmed. Culture studies using CSF showed no evidence of bacterial, tuberculosis, or fungal infection.
Spinal computed tomography (CT) and MRI scans were performed. The sagittal CT image revealed no abnormal bony structures and calcification lesion. Diffuse ventral and dorsal aspects of the spinal dura thickening through C2 to C5 levels were found on MRI scans. Low signal intensity was shown in the T1- and T2-weighted sequences, and relatively homogenous contrast enhancement with gadolinium was observed. Cord signal change and compressive myelopathy through C3 to C5 levels were confirmed (Fig. 1). Brain MRI was performed, and no findings suggestive of idiopathic hypertrophic cerebral pachymeningitis were confirmed.
MRI confirmed compressive myelopathy, and since the patientâs symptoms worsened, cervical decompressive laminectomy and laminoplasty at C2 to C5 were performed immediately. The operative field showed a yellow ligament with severe adhesion to the dura mater after laminectomy. Lesions and normal dura were carefully peeled off, and parts of the ligaments and outer dura mater thickened by chronic inflammation were removed. No complications such as CSF leakage or nerve damage occurred during surgery.
The pathologic findings confirmed degenerated fibrous tissue with mixed inflammatory cell infiltration, and ruled out the possibility of other diseases, including tumors. Immediately after surgery, the patientâs symptoms improved to motor grade III in the bilateral hand grasp evaluation and grade IV in the lower-extremity evaluation. He also showed subjective improvement of hypoesthesia and numbness under the T1 dermatome level. After surgery, the patient started taking oral prednisolone 60 mg once daily from postoperative day 10. After caring for the surgical wounds, the patient was transferred to the department of rehabilitation for rehabilitation treatment. After 2 months of oral steroid treatment, the prednisolone dose was reduced to 40 mg once daily and maintained at that level. The dose will be gradually tapered on the basis of the patientâs symptoms.
Rheumatoid factor, antinuclear antibody, double-stranded DNA, antiphospholipid antibody, lupus anticoagulant, anti-neutrophil cytoplasmic antibodies (p-ANCA and c-ANCA), Ro (SS-A), La (SS-B), U1RNP, Jo-1, and anti-cyclic citrullinated peptide levels were performed after discharge and noted within normal range. Follow-up cervical MRI was performed at one month after surgery, and radiological improvement was confirmed (Fig. 2). Five months after surgery, the patient complained of subjective weakness in the lower extremities, and cervical MRI was performed for follow-up evaluation. No significant radiological deterioration of the lesion was observed on MRI (Fig. 3). Up to 7 months after decompressive laminoplasty and sufficient steroid treatment, the patientâs performance gradually improved despite some minor disabilities.
IHSP is a rare disease that is known to cause thickening of the dura mater and arachnoid region due to chronic inflammation, mainly affecting the cervical and thoracic spinal cord level. The epidemiology of IHSP has not been clearly established, but some studies show that it usually affects people between 50 and 70 and is more common in women than men [10]. The symptoms of IHSP appear differently depending on the location and severity of the disease. The stage-based symptoms of IHSP were first reported by Charcot [11]. The first stage primarily shows progressive radicular symptoms, while the second stage shows muscle weakness and atrophy, and the third stage shows paralysis of the lower extremities and bladder and progressive anal dysfunction [12].
The etiology of IHSP is unclear. Research on its associations with various diseases is in progress, and studies have described associations with rheumatoid arthritis, Wegenerâs granulomatosis, sarcoidosis, metabolic diseases, autoimmune diseases, and transverse myelitis [13-15]. In our case, the patient had severe symptoms and showed compressive myelopathy on MRI. Surgery was performed prior to examination for related diseases, and diseases such as malignancies and tuberculosis were excluded from the biopsy.
Identification of patientsâ symptoms and distinction from symptoms caused by brain lesions is important. The recommended imaging modality for diagnosis is spinal MRI with gadolinium enhancement [6]. Since hypertrophy of the dura mater is a characteristic MRI finding, it is necessary to differentiate it from conditions such as an epidural abscess or spinal tumor. IHSP lesions are observed to be hypointense in T1- and T2-weighted images due to the characteristics of the dense fibrous tissue component of the lesion [16]. In our case, the lesion appeared as a typically diffuse thickened dura mater with homogenous enhancement. The gold standard for diagnosing IHSP is biopsy of the thickened dura mater [17]. Chronic nonspecific and granulomatous inflammation can be identified, and epidural abscess and tumorous conditions and diseases can be ruled out on the basis of the pathological and gross findings observed during surgery [16].
The gold standard treatment for IHSP remains controversial. In patients with neurological symptoms due to cord compression, surgical decompression and open biopsy are helpful for both treatment and diagnosis [18]. Because most of the lesions are extensive and long, complete or gross total resection of the lesion is impossible. The indications for surgical decompression and the extent of surgical resection of dural hypertrophy are topics of debate. Medical treatment mainly involves immunosuppressive therapy using steroid agents, but there is no clear consensus on the dose and duration of steroid therapy [9,17]. In addition to steroid therapy, some studies have also suggested radiation therapy, methotrexate, cyclophosphamide, and empirical anti-tuberculosis therapy [17]. Surgical treatment for pathologic diagnosis and decompression of the spinal cord and subsequent steroid therapy is the primary therapy at present [19]. Some studies recommend the additional use of methotrexate or azathioprine in patients who show relapse during steroid therapy or whose symptoms worsen after steroid tapering [8]. In our case, the patient showed an improvement in clinical and imaging studies up to 7 months after decompressive surgery and was only taking steroids continuously.
With sufficient medical steroid treatment and rehabilitation treatment, the prognosis of patients with IHSP can be improved even if only decompressive laminectomy and laminoplasty, without complications such as dural tears, are performed instead of aggressive surgery to remove the thickened ligament and dura mater extensively.
Fig. 1.
Preoperative sagittal spinal imaging (A) T2-weighted magnetic resonance imaging (MRI), (B) contrast-enhanced T1-weighted MRI, and (C) cervical computed tomography. T2-weighted axial cervical MRI obtained at the (D) C2, (E) C3, (F) C4, and (G) C5 levels. Written informed consent was obtained for publication of this case report and accompanying images.
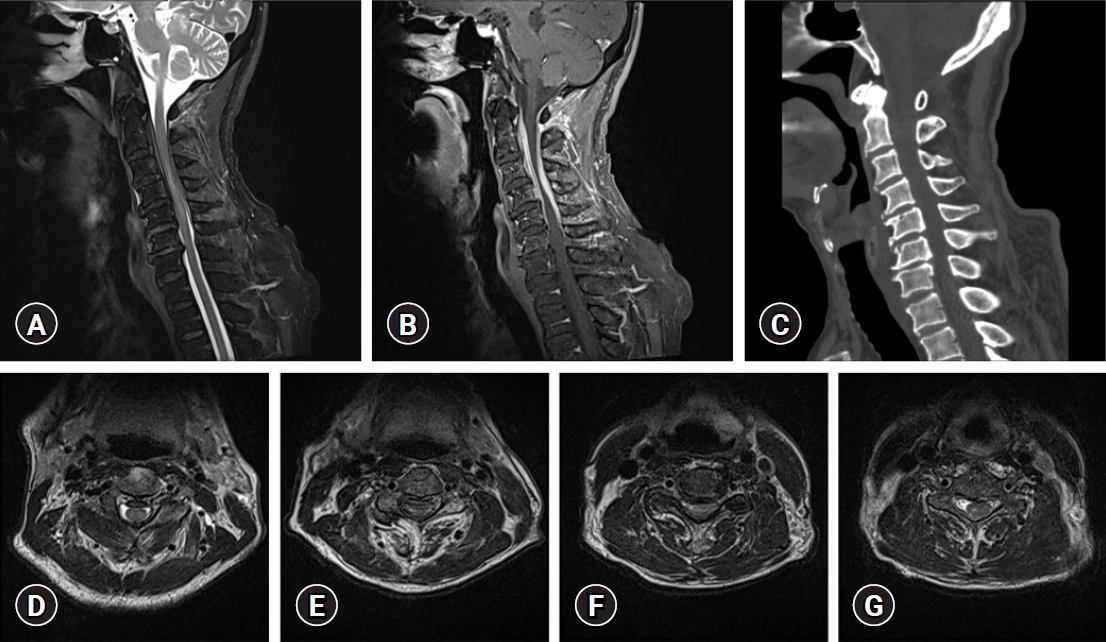
Fig. 2.
Sagittal spinal magnetic resonance imaging (MRI) obtained 1 month after the operation: (A) T2-weighted and (B) contrast-enhanced T1-weighted. T2-weighted axial cervical MRI obtained before surgery at the (C) C2, (D) C3, (E) C4, and (F) C5 levels. These images revealed that the lesion area was significantly thinner than on the preoperative MRI scans. Written informed consent was obtained for publication of this case report and accompanying images.

REFERENCES
1. Pai S, Welsh CT, Patel S, Rumboldt Z. Idiopathic hypertrophic spinal pachymeningitis: report of two cases with typical MR imaging findings. AJNR Am J Neuroradiol 2007;28:590-2.



3. Jee TK, Lee SH, Kim ES, Eoh W. Idiopathic hypertrophic spinal pachymeningitis with an osteolytic lesion. J Korean Neurosurg Soc 2014;56:162-5.


4. Park JY, Choi I, Khil EK, Kim WJ, Shin IY. Idiopathic hypertrophic spinal pachymeningitis with spinal cord lesion: a case report. Korean J Neurotrauma 2020;16:367-73.


5. Kanamori M, Matsui H, Terahata N, Tsuji H. Hypertrophic spinal pachymeningitis: a case report. Spine (Phila Pa 1976) 1997;22:1787-90.



6. Friedman DP, Flanders AE. Enhanced MR imaging of hypertrophic pachymeningitis. AJR Am J Roentgenol 1997;169:1425-8.


7. Park SH, Whang CJ, Sohn M, Oh YC, Lee CH, Whang YJ. Idiopathic hypertrophic spinal pachymeningitis: a case report. J Korean Med Sci 2001;16:683-8.


8. Kupersmith MJ, Martin V, Heller G, Shah A, Mitnick HJ. Idiopathic hypertrophic pachymeningitis. Neurology 2004;62:686-94.


9. Ranasinghe MG, Zalatimo O, Rizk E, et al. Idiopathic hypertrophic spinal pachymeningitis. J Neurosurg Spine 2011;15:195-201.


10. Olubajo F, Yermakova T, Highley JR, Arzoglou V. Concomitant idiopathic hypertrophic spinal pachymeningitis and Guillain-BarrĂŠ syndrome in a patient: coincidence or a triggering mechanism? J Neurosurg Spine 2017;27:335-40.


11. Charcot JM. Deux cas d'atrophie musculaire progressive: avec lÊsions de la substance grise et des faisceaux antÊrolatÊraux de la moelle Êpinière. Arch Physiol Norm Pathol 1869;2:354-69.
12. Park TJ, Seo WD, Kim SY, Cho JH, Kim DH, Kim KH. Effective response of methotrexate for recurrent idiopathic hypertrophic spinal pachymeningitis. Korean J Spine 2016;13:200-3.



14. Nishino H, Rubino FA, Parisi JE. The spectrum of neurologic involvement in Wegenerâs granulomatosis. Neurology 1993;43:1334-7.


15. Shu J, Gu C, Ren Y, Wei W. ANCA-associated hypertrophic spinal pachymeningitis presenting with longitudinally extensive transverse myelitis: a case report. Ann Indian Acad Neurol 2020;23:131-2.



16. Nishizaki T, Iwamoto F, Uesugi S, Akimura T, Yamashita K, Ito H. Idiopathic cranial pachymeningoencephalitis focally affecting the parietal dura mater and adjacent brain parenchyma: case report. Neurosurgery 1997;40:840-3.


17. Qin LX, Wang CY, Hu ZP, Zeng LW, Tan LM, Zhang HN. Idiopathic hypertrophic spinal pachymeningitis: a case report and review of literature. Eur Spine J 2015;24 Suppl 4:S636-43.


-
METRICS

-
- 0 Crossref
- 0 Scopus
- 2,160 View
- 24 Download
- ORCID iDs
-
Tae Hwan Shin

https://orcid.org/0000-0002-6607-661XTae Woo Kim

https://orcid.org/0000-0002-7457-8804 - Related articles




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Download Citation
Download Citation Print
Print